The
Metallurgy Of Carbon Steel
|
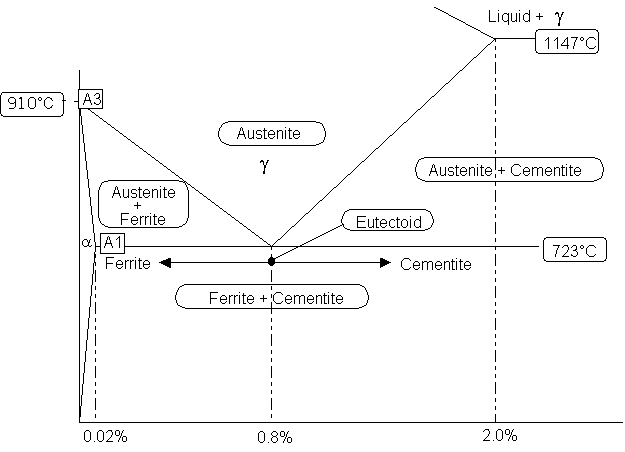
| The best way to understand the metallurgy of carbon steel is to study
the Iron Carbon Diagram. The diagram shown below is based on the
transformation that occurs as a result of slow heating. Slow cooling
will reduce the transformation temperatures; for example: the A1 point
would be reduced from 723°C to 690 °C. However the fast heating
and cooling rates encountered in welding will have a significant influence
on these temperatures, making the accurate prediction of weld metallurgy
using this diagram difficult. |
|
 |
-
Austenite This phase is only possible in carbon
steel at high temperature. It has a Face Centre Cubic (F.C.C) atomic
structure which can contain up to 2% carbon in solution.
-
Ferrite This phase has a Body Centre Cubic structure
(B.C.C) which can hold very little carbon; typically 0.0001% at room temperature.
It can exist as either: alpha or delta ferrite.
-
Carbon A very small interstitial atom that tends to
fit into clusters of iron atoms. It strengthens steel and gives it
the ability to harden by heat treatment. It also causes major problems
for welding , particularly if it exceeds 0.25% as it creates a hard microstructure
that is susceptible to hydrogen cracking. Carbon forms compounds
with other elements called carbides. Iron Carbide, Chrome Carbide
etc.
-
Cementite Unlike ferrite and austenite, cementite is
a very hard intermetallic compound consisting of 6.7% carbon and the remainder
iron, its chemical symbol is Fe3C. Cementite is very hard,
but when mixed with soft ferrite layers its average hardness is reduced
considerably. Slow cooling gives course perlite; soft easy to machine but
poor toughness. Faster cooling gives very fine layers of ferrite
and cementite; harder and tougher
-
Pearlite A mixture of alternate strips of ferrite and
cementite in a single grain. The distance between the plates and
their thickness is dependant on the cooling rate of the material;
fast cooling creates thin plates that are close together and slow cooling
creates a much coarser structure possessing less toughness. The name
for this structure is derived from its mother of pearl appearance under
a microscope. A fully pearlitic structure occurs at 0.8% Carbon.
Further increases in carbon will create cementite at the grain boundaries,
which will start to weaken the steel.
|
 |
-
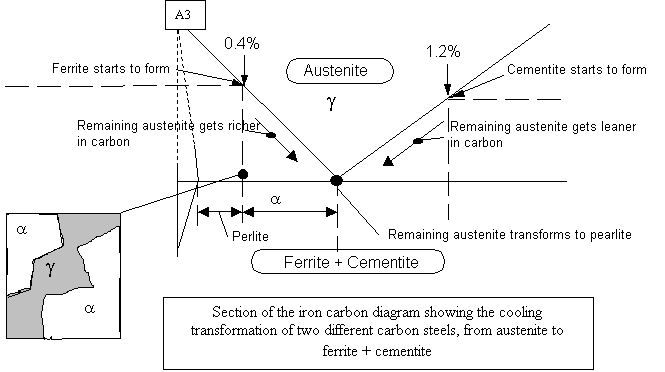
Cooling of a steel below 0.8% carbon When a steel
solidifies it forms austenite. When the temperature falls below the
A3 point, grains of ferrite start to form. As more grains of ferrite
start to form the remaining austenite becomes richer in carbon. At
about 723°C the remaining austenite, which now contains 0.8% carbon,
changes to pearlite. The resulting structure is a mixture consisting
of white grains of ferrite mixed with darker grains of pearlite.
Heating is basically the same thing in reverse.
|
|
 |
-
Martensite If steel is cooled rapidly from austenite,
the F.C.C structure rapidly changes to B.C.C leaving insufficient time
for the carbon to form pearlite. This results in a distorted structure
that has the appearance of fine needles. There is no partial transformation
associated with martensite, it either forms or it doesnt. However,
only the parts of a section that cool fast enough will form martensite;
in a thick section it will only form to a certain depth, and if the shape
is complex it may only form in small pockets. The hardness
of martensite is solely dependant on carbon content, it is normally very
high, unless the carbon content is exceptionally low.
-
Tempering The carbon trapped in the martensite transformation
can be released by heating the steel below the A1 transformation temperature.
This release of carbon from nucleated areas allows the structure to deform
plastically and relive some of its internal stresses. This reduces hardness
and increases toughness, but it also tends to reduce tensile strength.
The degree of tempering is dependant on temperature and time; temperature
having the greatest influence.
-
Annealing This term is often used to define a heat
treatment process that produces some softening of the structure.
True annealing involves heating the steel to austenite and holding for
some time to create a stable structure. The steel is then cooled
very slowly to room temperature. This produces a very soft structure,
but also creates very large grains, which are seldom desirable because
of poor toughness.
-
Normalising Returns the structure back to normal.
The steel is heated until it just starts to form austenite; it is then
cooled in air. This moderately rapid transformation creates relatively
fine grains with uniform pearlite.
-
Welding If the temperature profile for a typical weld
is plotted against the carbon equilibrium diagram, a wide variety of transformation
and heat treatments will be observed.
|
|
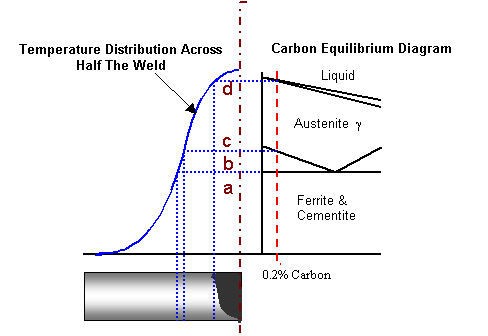 |
Note, the carbon equilibrium diagram shown above is only for
illustration, in reality it will be heavily distorted because of the rapid
heating and cooling rates involved in the welding process.
a)
b)
c)
d) |
Mixture of ferrite and pearlite grains; temperature below A1, therefore
microstructure not significantly affected.
Pearlite transformed to Austenite, but not sufficient temperature available
to exceed the A3 line, therefore not all ferrite grains transform to Austenite.
On cooling, only the transformed grains will be normalised.
Temperature just exceeds A3 line, full Austenite transformation.
On cooling all grains will be normalised
Temperature significantly exceeds A3 line permitting grains to grow.
On cooling, ferrite will form at the grain boundaries, and a course pearlite
will form inside the grains. A course grain structure is more readily
hardened than a finer one, therefore if the cooling rate between 800°C
to 500°C is rapid, a hard microstructure will be formed. This
is why a brittle fracture is most likely to propagate in this region.
|
-
Welds The metallurgy of a weld is very different from the
parent material. Welding filler metals are designed to create strong
and tough welds, they contain fine oxide particles that permit the nucleation
of fine grains. When a weld solidifies, its grains grow from the
course HAZ grain structure, further refinement takes place within these
course grains creating the typical acicular ferrite formation shown opposite.
|
 |
|
|
Recommended Reading
-
Metals and How To Weld Them :- Lincoln Arc Foundation
A very cheap hard backed book covering all the basic essentials of
welding metallurgy.
Welding Metallurgy Training Modules:- (Devised by The Welding
Institute of Canada) Published in the UK by Abington Publishing.
Not cheap but the information is clearly represented in a very readable
format.
|
|
Return To Sub Menu
Page last updated 08 May 2002 |